A BSI Certified Brand That You Can Trust.
Accurate Meditech Inc. is an ISO 13485 certified medical equipment design house and manufacturer dedicated in bringing you the best portable and easy to use vital sign monitoring solutions.







Trusted Worldwide
Delivering reliable vital sign monitoring solutions across the globe.
Our solutions are trusted by partners and users across the United States, Europe, the Middle East, Southeast Asia, and beyond.
A Coin Size Blood Pressure Monitor, the Last Piece of Apple Watch
Website: https://www.dotbglobal.com/trade-show/discover-company-ces
Accurate Meditech Inc. has developed a sensor hub that continuously measures blood pressure without requiring an inflatable cuff. It enables users to measure and track blood pressure conveniently as they go about their daily activities, bringing a new way of monitoring one's cardiovascular health.
In June 2023, it passed the review of the US FDA for the first time and obtained the world's first OTC cuffless blood pressure monitor.
This is a significant breakthrough in the history of ACCURATE and blood pressure monitors. However, we don't let that stop us.
While many smartwatches occupy various markets, the blood pressure function has not been realized by companies such as APPLE and Garmin.
ACCURATE, as the leader of cuffless, has a 510K license and has even developed a blood pressure measurement device, it is not easy to replace the Apple watch.
However, as the first and only one on the market that has obtained an OTC license, it has reduced the blood pressure monitor to the size of a coin and combined it with the strap of the APPLE Watch. It is an independently operating device with a precise algorithm, and the results are presented on the computer through Bluetooth on the APPLE Watch.
For the user, the original usage habits will not change. The only thing to do is to change the watch strap to a microCare-specific strap (the same as the commercially available design). Whenever and wherever put microCare into the unique strap and add Blood Pressure to your Apple Health.

History
Accurate Meditech Inc., was found in Taipei Taiwan in May 2017 by Stanley Wang, a veteran in both medical technologies and consumer electronics industries, with a product idea in creating a sticky patch sensor that reads blood pressure. After gaining precious experiences from collaborations with several well-established electronics corporates end in 2019, the company started to assemble an in-house team of experts in multiple fields, and shift its primary focus onto developing wearable non-invasive calibration-free physiological monitors. In 2022, while the first product, the Accurate Mini, enters working prototype stage, the company is also certified by BSI for ISO 13485.
Advisory Team

Tony Guo
Senior Technical Consultant

Douglas E. Herrington
Medical Device Consultant

INNOVATUS LAW
Legal Advisor
Compliance

Our devices meet the highest international regulatory standards for safety, accuracy, and reliability.





Certifications

Recognised and approved by leading regulatory authorities across multiple countries and regions.






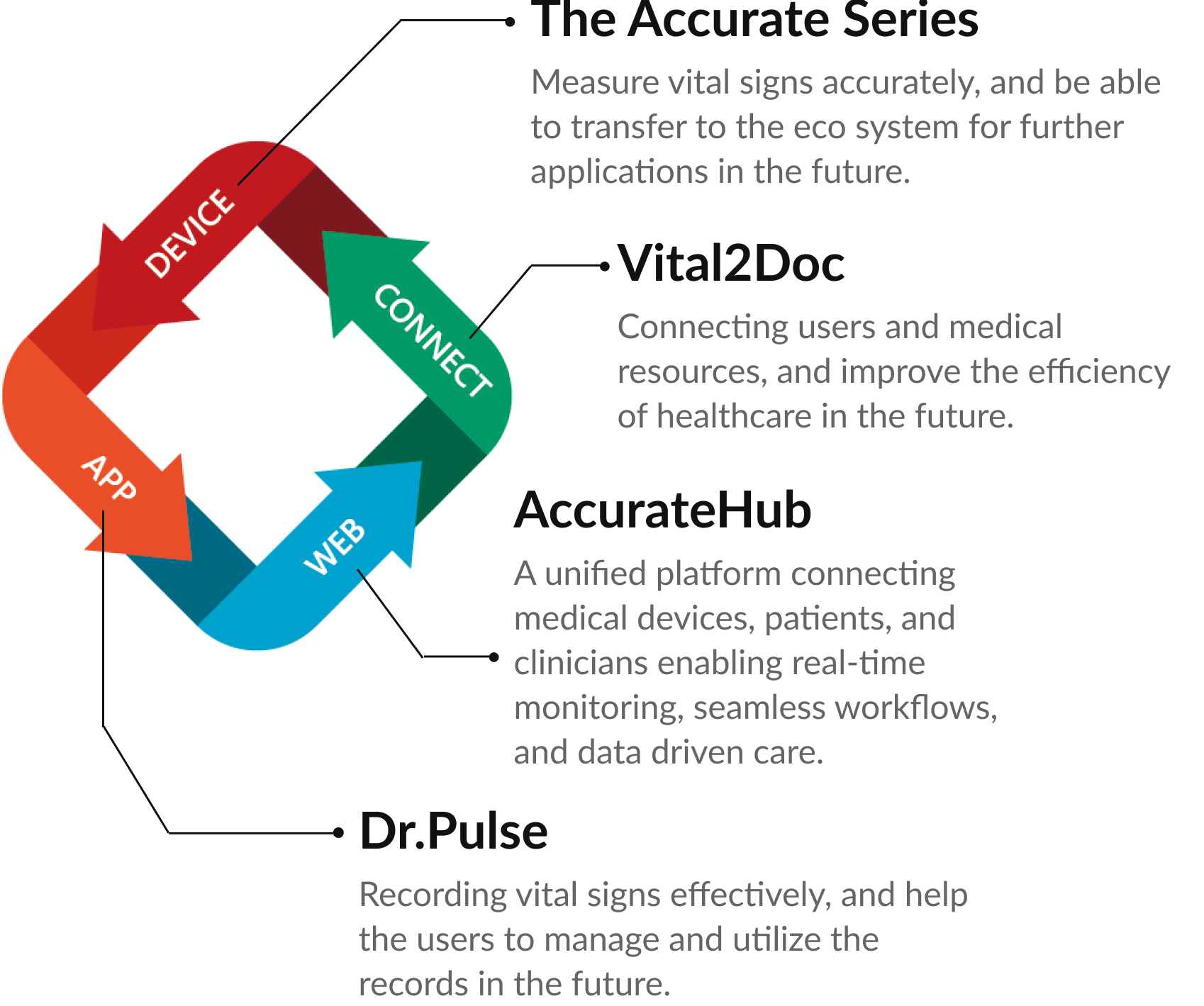
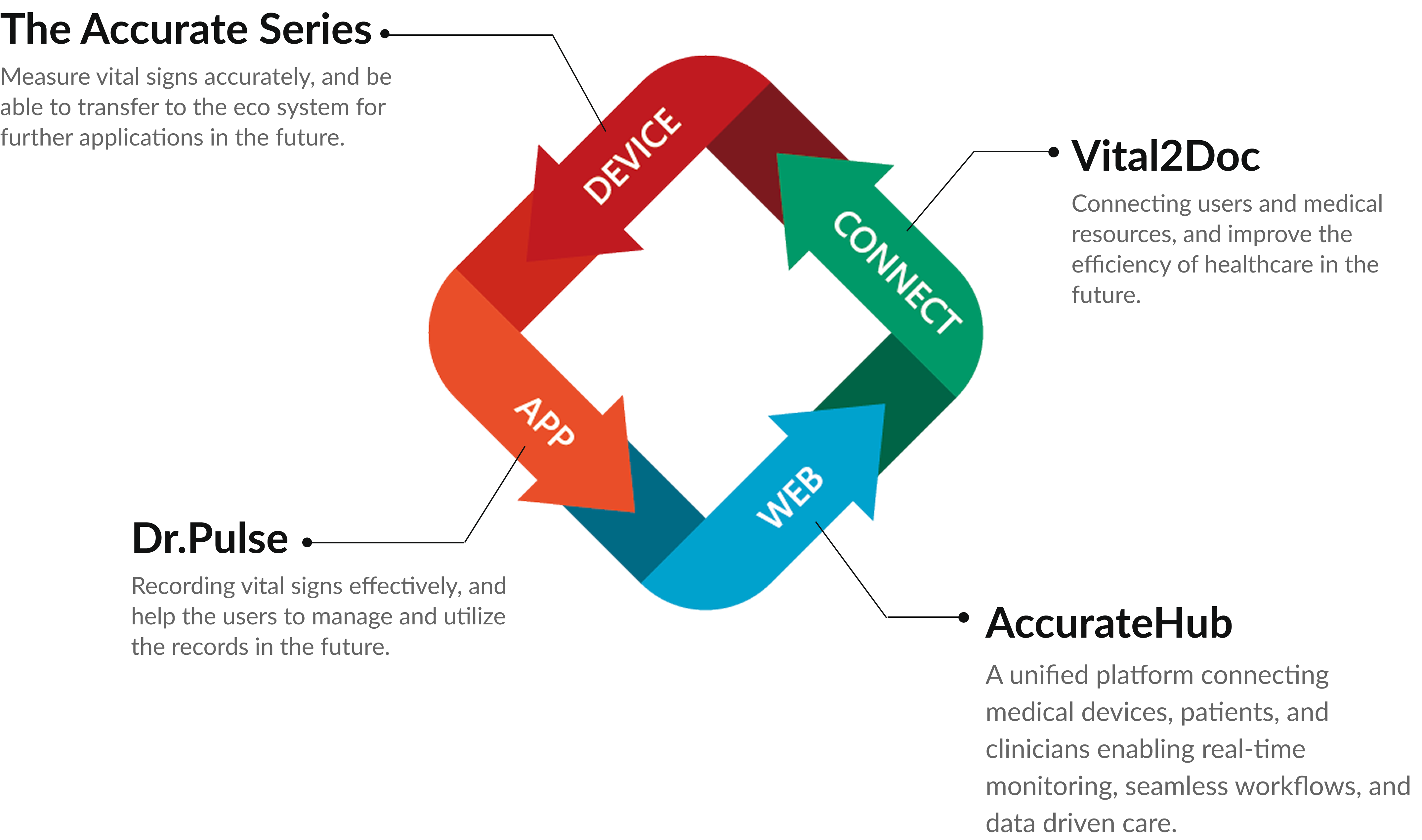
Eco System
Delivering reliable vital sign monitoring solutions across the globe.