Technology Backed by Scientific Studies
Information, researches, scientific papers and clinical studies from international health agencies and accredited institutions.

Clinical Trials

The most critical step to validate the performance, useability and safety of medical devices with international standards recognized by U.S. FDA., and eliminate possible issues before the release of our products.
International Standards Complied
ISO 81060-2: 2018
Demographics and characteristics
We covered multiple user groups include but not limited to: gender, age, and blood pressure.

Reference Sphygmomanometer
Welch Allyn DuraShock DS66 Trigger Aneroid
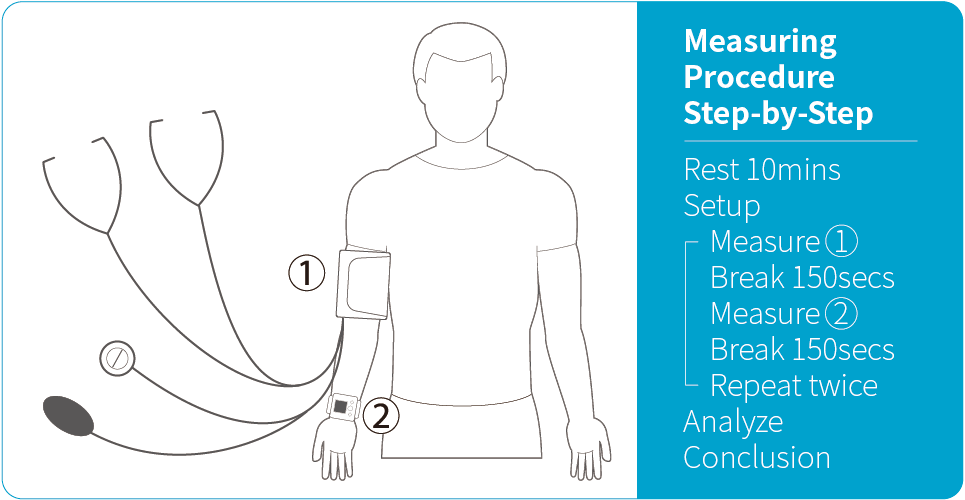
Measuring Procedure
The procedure used in this study follows ISO 81060-2:2018 Clause 5.2.4.1.1. By compare results of reference and subject device using sequential method repeatedly to gain 3 sets of valid readings from the same arm of the same patient to minimal external interferes and change in the patient's physiological condition. In order to be considered as a valid set of blood pressure readings, the differences between the reference and our device should be within 12 mmHg for systolic, and within 8 mmHg for diastolic.
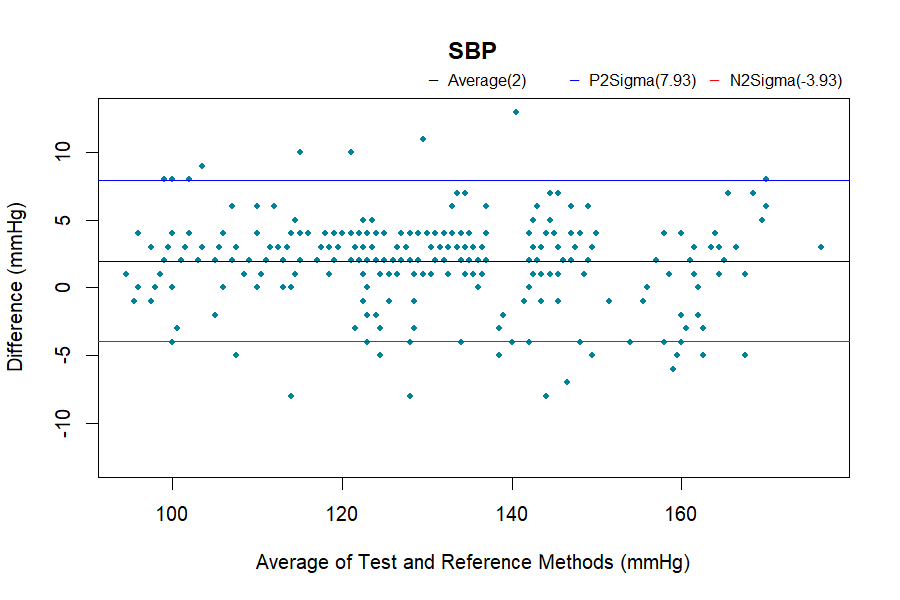
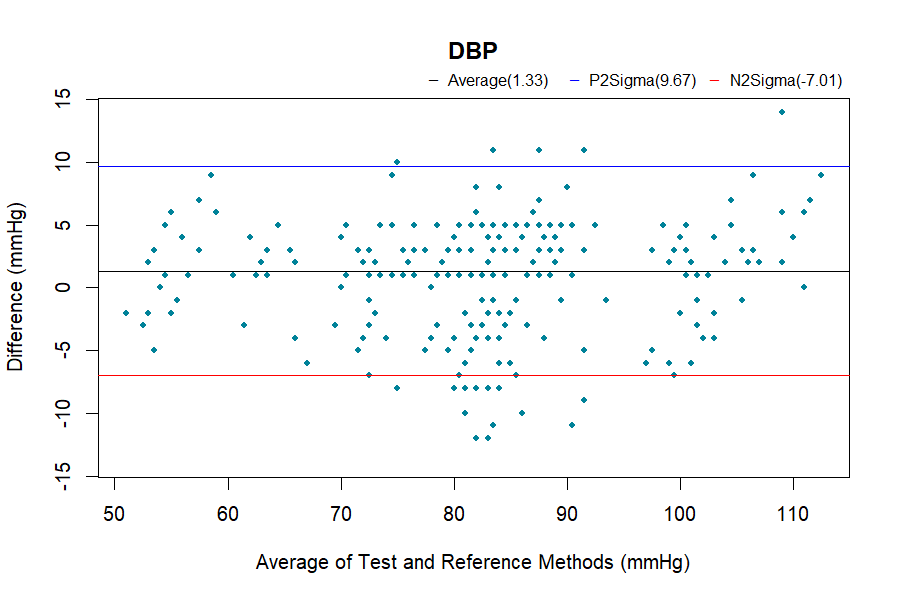
Analysis Methods
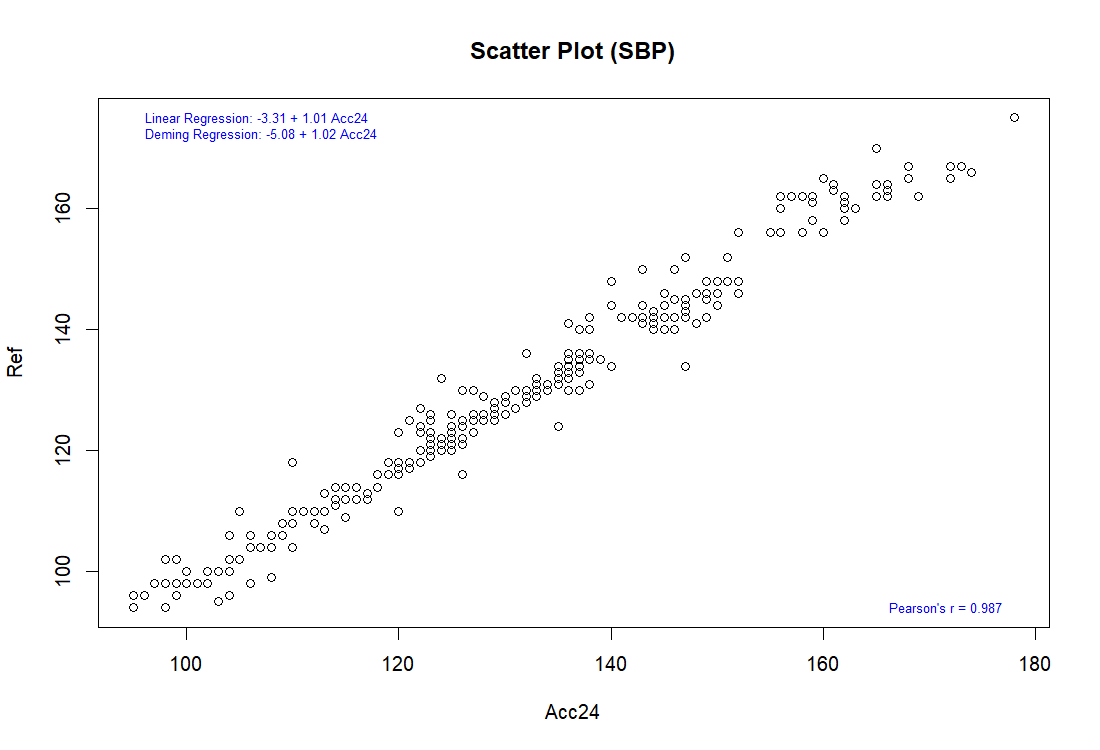
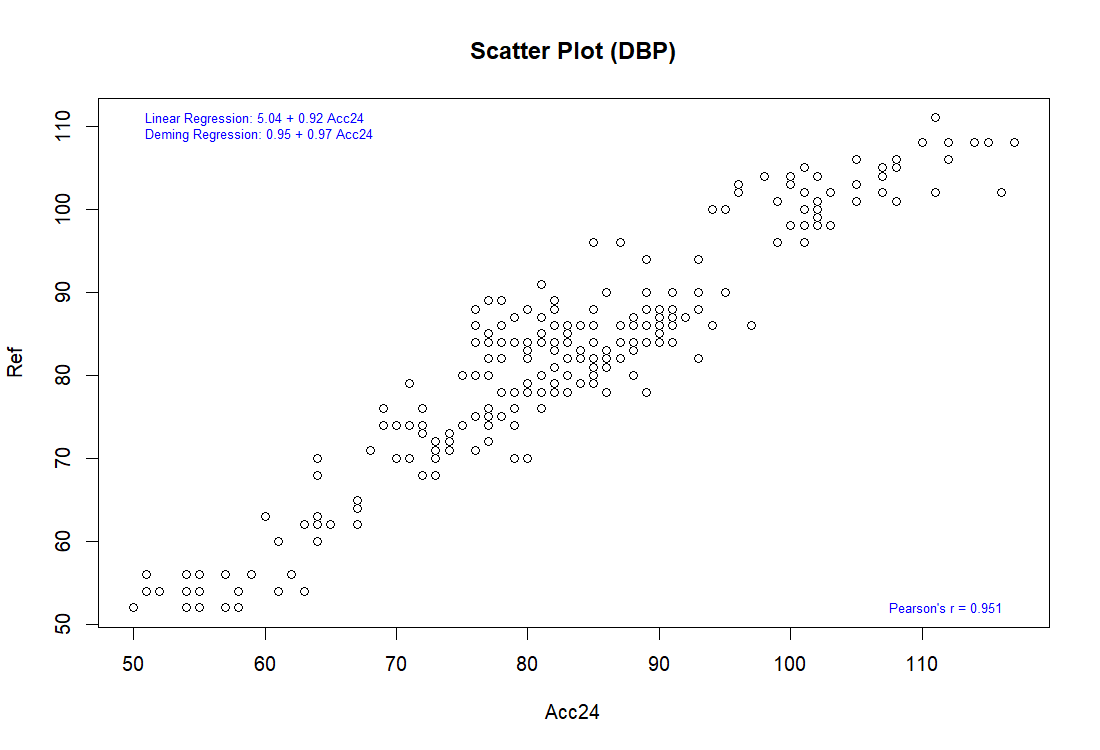
Bland-Altman analysis
Pearson correlation
Passing Criteria
The complete validation procedures should be conducted in at least total of 85 subjects, as specified in the ISO 81060-2 standards, with at least 255 BP determinations having a mean error magnitude ≤ 5 mmHg and a standard deviation ≤ 8 mmHg.
Results
General Group: Pass
Specialty Group: Pass